Background to Mirus Bio Virusgen transfection reagent
Gene therapies have garnered attention as an exciting treatment strategy for diseases that have a strong underlying genetic element. A sizeable percentage of these gene therapies are utilising either Adeno-associated virus (AAV) or lentiviruses (LV) as a delivery mechanism for the delivery of genetic material into target cells for various purposes, such as gene expression, gene knockout, or gene therapy.
This particular method sets itself apart from other technologies due to its non-integrating properties, ability to infect a broad range of cell types, and its safety profile. There are currently a plethora of AAV and LV treatments in pre-clinical and clinical stages of development. One of the key AAV therapies on the market today is Luxturna, a treatment designed for individuals with loss of vision due to inherited retinal dystrophy.
The process of generating AAVs involves 3 key steps;
- Plasmid construction: create a plasmid containing the AAV inverted terminal repeats, the actual gene of interest and the necessary packaging genes.
- Transfection: transfect a producer cell line such as HEK293 with the AAV plasmid containing the gene of interest
- Harvesting: collecting and purifying the AAV particles from the culture medium.
Despite this there are still several issues facing the widespread use of AAVs and LVs as a treatment modality for certain genetic disorders. These include:
- Low yield AAV vectors: Achieving high viral vector yields can be challenging. Low transfection efficiency, poor replication of AAV in producer cells, or issues with the helper plasmids can contribute to low yields.
- Cell line productivity: The choice of the producer cell line is crucial. Some cell lines may not support efficient AAV replication or packaging. HEK293 cells are commonly used, but variations in subtypes (e.g., HEK293T) and passage number can impact productivity.
- Purification challenges: Purifying AAV particles from cellular contaminants can be difficult. Common purification methods involve ultracentrifugation, chromatography, or precipitation, each with its own challenges and limitations.
MSC has partnered with Mirus bio, a company founded in 1995 by the Internationally recognised molecular biologist Jon Wolf in University of Wisconsin-Madison. To date they have more than 40000 users globally, over 10000 publications and their products have been tested in over 1200 cell lines. Since its founding Mirus Bio has been meeting industry and customer needs through innovation, collaboration and partnerships.
Mirus Bio TransIT-VirusGEN® GMP Transfection Reagent is designed to enhance delivery of packaging and transfer vector DNA to suspension and adherent HEK 293 cell types in order to increase production of recombinant lentivirus and adeno-associated virus (AAV)
Why choose Mirus’s TransIT-VirusGEN® Transfection Reagent?
Key benefits:
- Performance – Efficient DNA delivery for production of high-titer viral vectors.
- Scalability – Efficient production from < 1 ml to large scale cultures.
- Flexibility – Compatible with different recombinant virus and cell culture systems.
- Animal Origin Free – Fully synthetic transfection reagent formulation.
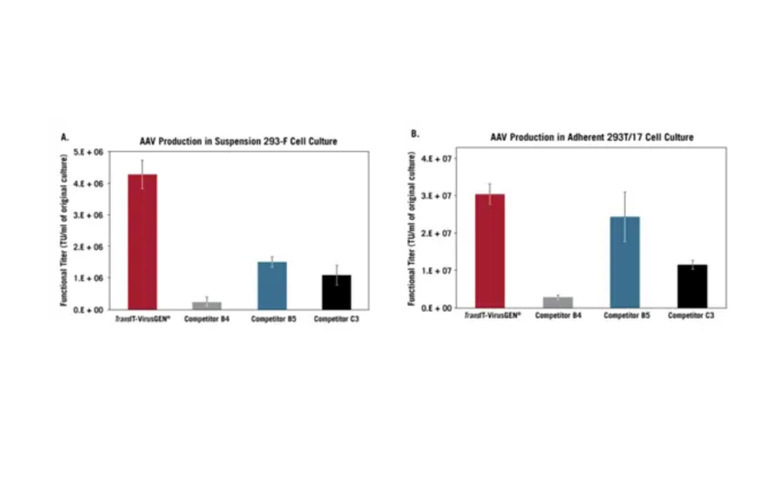
TransIT-VirusGEN® Outperforms Competitor Reagents in Suspension & Adherent AAV Cell Cultures. Suspension FreeStyle™ 293-F cells grown in FreeStyle™ F17 Medium (A) or adherent 293T/17 cells (B) were transfected with pAAV-hrGFP, pAAV-RC, and pAAV-Helper (1:1:1 ratio, 1.5 µg/ml, Agilent Technologies) with the following reagents: TransIT-VirusGEN® (2:1, vol:wt), Competitor B4 (3:1), Competitor B5 (3:3:1) or 25 kDa PEI (4:1, PolySciences). Harvested virus was used to transduce HT1080 cells and GFP expression was measured 48 hours post-transduction using guava easyCyte™ 5HT Flow Cytometer. Functional titers were measured from virus dilutions with less than 20% GFP positive cells. The error bars represent the standard deviation of triplicate wells.
Get In Touch


